
We are currently facing a silent epidemic known as Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). This condition is the most common cause of chronic liver disease globally. In the United States it affects approximately 1 in 3-4 adults1. The prevalence of MASLD continues to rise alongside the growing obesity and diabetes epidemic2. Alarmingly, most individuals with MASLD are unaware of their condition, as it often presents with no symptoms3.
What is MASLD?
MASLD is a complex metabolic disorder influenced by multiple factors and is part of a broader systemic disease. Diagnosis is based on the evidence of steatosis (fat accumulation in the liver) seen on imaging or biopsy, along with the presence of at least one of five cardiometabolic risk factors. These risk factors include ethnicity-adjusted Body Mass Index (BMI) or waist circumference, insulin resistance or diabetes, untreated or treated hypertension, hypertriglyceridemia, or dyslipidemia (abnormal lipid levels), or treatment with a lipid-lowering agent4.
Although less than 10% of individuals with MASLD will develop liver-related complications, the primary causes of death in these patients are cardiovascular issues and non-liver-related cancers5. About 25% of patients with MASLD progress to a more severe form called steatohepatitis (MASH), which causes inflammation and injury to the liver. If left untreated, approximately 25% of those with steatohepatitis may develop irreversible liver damage, leading to cirrhosis. Identifying patients with fibrosis is crucial to assessing prognosis, as all-cause and liver-specific mortality rates significantly increase in patients with stage 2 fibrosis or higher6. Therefore, those with steatohepatitis and F2 fibrosis are often categorized as “at-risk MASH.”
Who Should Be Screened for MASLD?
Currently, there are no recommendations for routine screening due to uncertainties surrounding testing, treatments, long-term benefits, and cost-effectiveness. There should be a heightened awareness of MASLD and MASH among individuals with type 2 diabetes, obesity, or metabolic syndrome, as these populations have a higher prevalence of the disease.
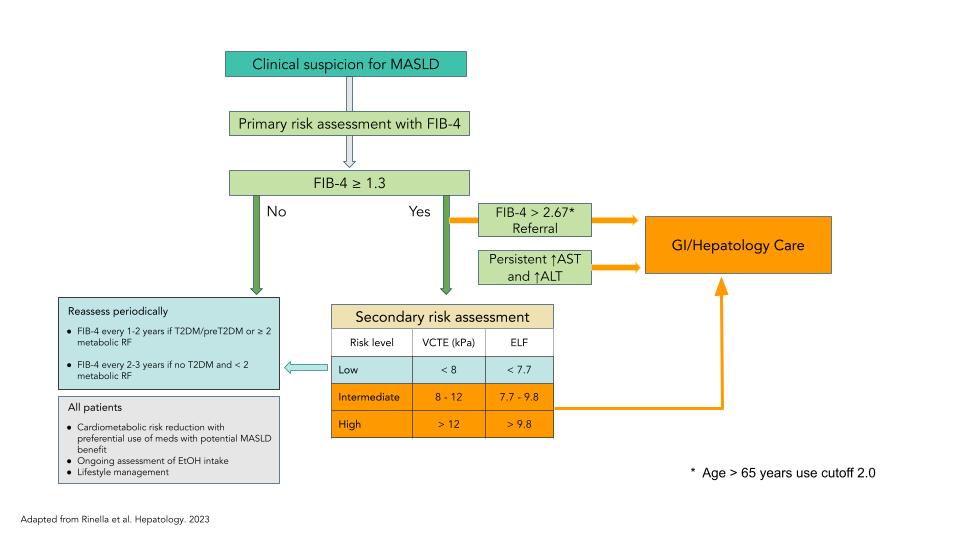
After excluding secondary causes of steatosis, a primary risk assessment can be conducted using the FIB-4 score. This score is calculated from the patient’s age, AST and ALT levels, and platelet count, assisting in stratifying patients into low, intermediate, or high likelihood of advanced fibrosis. A FIB-4 score of less than 1.3 is highly predictive for ruling out advanced fibrosis, allowing for management in primary care with a focus on reducing cardiometabolic risks. Conversely, a FIB-4 score of 2.67 or higher is strongly predictive of advanced fibrosis, warranting a referral to gastroenterology/hepatology for further evaluation and potential liver-directed treatments (the cutoff should be adjusted to 2 for patients over 65 years of age).
For those with indeterminate scores between 1.3 and 2.67, additional risk assessment is advised, using non-invasive testing such as vibration-controlled transient elastography (VCTE) or Fibroscan, or blood tests like Enhanced Liver Fibrosis (ELF). Patients with low-risk results from secondary testing can continue to be managed in primary care, while those with indeterminate or high scores should be referred to a specialist. The FIB-4 score should be reassessed every 1-2 years for individuals with pre-diabetes/diabetes or more than two metabolic risk factors. In the absence of pre-diabetes or diabetes and with fewer than two metabolic risk factors, the FIB-4 score can be monitored every 2-3 years (Figure 1)4,7.
How Should Patients with MASLD Be Managed?
Lifestyle management is the cornerstone of treatment for individuals with MASLD. A diet high in fiber, whole foods, and low in sugar (especially fructose and processed beverages) is recommended. The Mediterranean diet is a good option due to its proven cardiometabolic benefits7,8. While the specific macronutrient composition of the diet is important, sustained weight loss appears to be the most significant factor for improvement. Weight loss of 3-5% is necessary to enhance liver steatosis, while a greater weight loss of 7-10% is required to improve the histologic features of MASH and potentially fibrosis. Engaging in exercise for more than 150 minutes per week can improve liver lipid content and reduce cardiovascular risk, regardless of weight changes.
All patients with MASLD should have their cardiometabolic risks addressed and should be prescribed medications that may benefit MASLD. Additionally, alcohol intake should be evaluated as a potential aggravating factor, especially for those at risk for MASH4,7,8.
Recently, two medications received conditional approval for the treatment of MASH with F2 or F3 fibrosis. The first medication to receive approval is Resmetirom, a liver-directed thyroid hormone receptor-β agonist (THR-β). Resmetirom binds to receptors in the nucleus of liver cells, increasing genes involved in lipid metabolism9. The second medication to receive conditional approval to treat MASH is Wegovy (semaglutide) injection, a glucagon-like peptide-1 receptor agonist (GLP-1)11. Wegovy is also FDA approved for treatment of obesity and cardiovascular risks12. However, patients with significant fibrosis should be managed in collaboration with a GI/hepatologist.
References:
- Younossi, Zobair M.1,2,3; Golabi, Pegah1,2,3; Paik, James M.1,2,3; Henry, Austin1; Van Dongen, Catherine1; Henry, Linda1,2,3. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology 77(4):p 1335-1347, April 2023. | DOI: 10.1097/HEP.0000000000000004
- Younossi ZM, Kalligeros M, Henry L. Epidemiology of metabolic dysfunction-associated steatotic liver disease. Clin Mol Hepatol. 2025;31(Suppl):S32-S50.
- Alqahtani, Saleh A.1,2,3; Paik, James M.4,5; Biswas, Rakesh4; Arshad, Tamoore4; Henry, Linda1; Younossi, Zobair M.*,4,5,6. Poor Awareness of Liver Disease Among Adults With NAFLD in the United States. Hepatology Communications 5(11):p 1833-1847, November 2021. | DOI: 10.1002/hep4.1765
- Rinella, Mary E.1; Neuschwander-Tetri, Brent A.2; Siddiqui, Mohammad Shadab3; Abdelmalek, Manal F.4; Caldwell, Stephen5; Barb, Diana6; Kleiner, David E.7; Loomba, Rohit8. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology 77(5):p 1797-1835, May 2023. | DOI: 10.1097/HEP.0000000000000323
- Stefan, N. et al. Metabolic dysfunction-associated steatotic liver disease: heterogenous pathomechanisms and effectiveness of metabolism-based treatment. The Lancet Diabetes-Endocrinology. Vol 13. Feb. 2025.
- Loomba, Rohit et al. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell, Volume 184, Issue 10, 2537 – 2564
- Kenneth Cusi, Manal F. Abdelmalek, Caroline M. Apovian, Kirthikaa Balapattabi, Raveendhara R. Bannuru, Diana Barb, Joan K. Bardsley, Elizabeth A. Beverly, Karen D. Corbin, Nuha A. ElSayed, Scott Isaacs, Fasiha Kanwal, Elizabeth J. Pekas, Caroline R. Richardson, Michael Roden, Arun J. Sanyal, Jay H. Shubrook, Zobair M. Younossi, Mandeep Bajaj; Metabolic Dysfunction–Associated Steatotic Liver Disease (MASLD) in People With Diabetes: The Need for Screening and Early Intervention. A Consensus Report of the American Diabetes Association. Diabetes Care 20 June 2025; 48 (7): 1057–1082. https://doi.org/10.2337/dci24-0094
- European Association for the Study of the Liver (EASL)European Association for the Study of Diabetes (EASD)European Association for the Study of Obesity (EASO) et al. EASL–EASD–EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). Journal of Hepatology, Volume 81, Issue 3, 492 – 542
- Harrison SA, Bedossa P, Guy CD, Schattenberg JM, Loomba R, Taub R, Labriola D, Moussa SE, Neff GW, Rinella ME, Anstee QM, Abdelmalek MF, Younossi Z, Baum SJ, Francque S, Charlton
- MR, Newsome PN, Lanthier N, Schiefke I, Mangia A, Pericàs JM, Patil R, Sanyal AJ, Noureddin M, Bansal MB, Alkhouri N, Castera L, Rudraraju M, Ratziu V; MAESTRO-NASH Investigators. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N Engl J Med. 2024 Feb 8;390(6):497-509. doi: 10.1056/NEJMoa2309000. PMID: 38324483.
- Sanyal AJ, Newsome PN, Kliers I, Østergaard LH, Long MT, Kjær MS, Cali AMG, Bugianesi E, Rinella ME, Roden M, Ratziu V; ESSENCE Study Group. Phase 3 Trial of Semaglutide in Metabolic Dysfunction-Associated Steatohepatitis. N Engl J Med. 2025 Jun 5;392(21):2089-2099. doi: 10.1056/NEJMoa2413258. Epub 2025 Apr 30. PMID: 40305708.
- Center for Drug Evaluation and Research. (n.d.). FDA approves treatment for serious liver disease known as “mash.” U.S. Food and Drug Administration. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-treatment-serious-liver-disease-known-mash